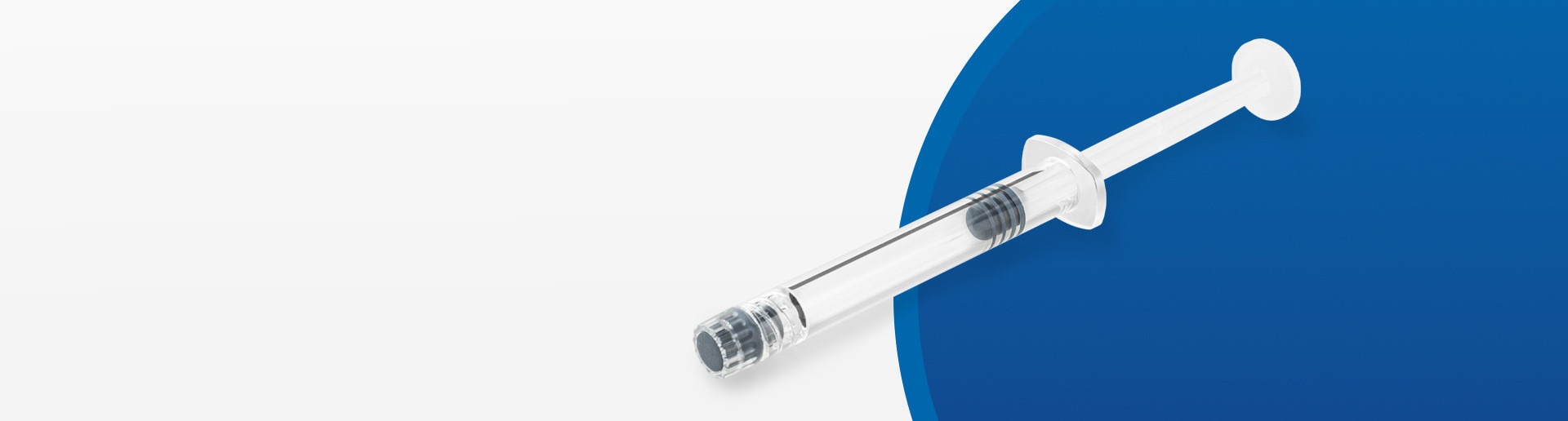
EZ-fill® Kit: Ready-to-use components for fast access to small volumes

All the advantages of the EZ-fill® platform in a high-quality compact packaging kit. A complete high-quality kit to meet your needs across the entire drug life cycle, from the clinical phase to small commercial batches. The perfect solution to protect, store, and deliver your drug product safely.
News & Press Releases
Discover allNews & Press Releases
Media Wall
Discover all
Stainless steel needles: quantifying cobalt risk to comply with eu medical device regulation
JAN 17 |
2024

Meeting market needs while mitigating time, risk and cost
JAN 16 |
2024


Innovating on-body delivery systems with Vertiva™
OCT 26 |
2023
Media Wall
JAN 17 |
2024

Stainless steel needles: quantifying cobalt risk to comply with eu medical device regulation
JAN 16 |
2024

Meeting market needs while mitigating time, risk and cost
OCT 26 |
2023

Innovating on-body delivery systems with Vertiva™
Upcoming Events & Webinars

DCAT Week 2024
18 - 21 March 2024
The Fifty Sonesta Select New York
New York, United States
DISCOVER MORE

DCAT Week 2024
18 - 21 March 2024
The Fifty Sonesta Select New York
New York, United States
DISCOVER MORE